How Are Essential Oils Extracted?
What Are Essential Oils?
 Essential oils are natural aromatic extracts from plant material including grasses, leaves, flowers, needles, twigs, peels of fruit, seeds, bark, roots, and underground stems (rhizomes).
Essential oils are natural aromatic extracts from plant material including grasses, leaves, flowers, needles, twigs, peels of fruit, seeds, bark, roots, and underground stems (rhizomes).
For example, rose essential oil comes from the flowers, basil from the leaves, lime from the rind, anise from the seeds, sandalwood from the wood, frankincense from the resin of its tree, ginger from from rhizomes, and so on.
The Merriam-Webster dictionary defines essential oils as, “any of a class of volatile oils that give plants their characteristic odors and are used especially in perfumes and flavorings, and for aromatherapy.”
The word "volatile" means a substance that easily evaporates at normal temperatures. These volatile oils given off by plants are responsible for the aromatic scents of flowers and other plant parts.
All citrus essential oils and others like fir needle and lavender are fairly volatile, while others like clove, cedarwood, patchouli, and sandalwood are less volatile.
These aromatic essential oil compounds are stored in tiny pockets in plant material and must be extracted to be released. The type of plant material (leaves, flowers, roots) being used determines which method of extraction will produce the best results.
For more detailed information please read our blog, "What Are Essential Oils."
Methods of Extraction
"True essential oils" are ONLY extracted either by steam distillation or, in the case of citrus oils, expression.
Distillation
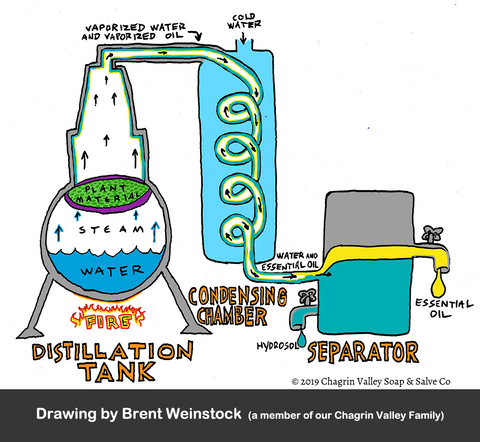 Steam distillation is the most commonly used method to extract essential oils.
Steam distillation is the most commonly used method to extract essential oils.
During this process the botanicals are placed into a still and pressurized steam is forced through the plant material--imagine using a steaming basket on your stove.
The hot steam forces open pockets of aromatic compounds. The compounds escape from the plant material and evaporate into the steam.
The steam must be hot enough to allow the release of the essential oil while not damaging the plant material. The steam along with the volatile essential oil (the plant that is able to convert into vapor) then passes through a cooling system where the steam condenses into a liquid consisting of essential oil and water.
The essential oil, being lighter than the water, will float to the top and can then be separated from the water. The water by-product of distillation is called a hydrosol or floral water.
Learn more: Read our blog, "What is a Hydrosol?"
Steam distillation is the best process of extracting essential oils because the volatile compounds can be distilled at temperatures that are lower than their boiling points. In this way, the natural qualities of the plant material are less likely to become altered or diminished during the process. For this reason, essential oil distillation is the best extraction method that allows for purity.
Expression
Expression is a cold, mechanical method used for extracting essential oils from the rinds of citrus fruits. Anyone who has peeled a fresh citrus fruit knows the refreshing, energizing aroma. But some citrus oils do not hold up well when heat is used in extraction so they are usually obtained by a method called expression.
Expression is also known as the "expeller-pressed" or "cold-pressed" method of extraction since no heat is needed to extract the essential oil. This extraction basically involves squeezing the plant material in order to release the plant’s natural oils.
It is mostly used to extract citrus essential oils. In this process, the peels are pricked in order to puncture the cells containing the oils.
The peels are soaked in warm water and the oil is forced from the material under high mechanical pressure. Many base or carrier oils are extracted in a similar way.
Steam distillation and expression are the only two methods used to obtain "true essential oils."
However, some aromatic scents come from botanicals for which expression will not work and they cannot tolerate high-heat steam distillation.
As a result, the aromatic compounds of many flowers, such as neroli (orange blossom), jasmine, and rose are obtained using solvents.
Concretes & Absolutes:
Concretes and Absolutes are similar to essential oils since they are highly aromatic, concentrated extracts from plants. However, while essential oils must be produced by steam distillation or expression, concretes and absolutes require the use of a solvent for extraction and therefore cannot be called essential oils.
Fresh flowers or other plant materials are treated with a chemical solvent (see below). The "aromatic molecules" of the plant material are dissolved by the solvent thus separating them from the plant matter. Once the solvent is removed what is left behind is a solid waxy aromatic compound called a concrete.
The waxes are separated out and the aromatic oils are extracted from the concrete using ethyl alcohol. Once the alcohol is removed, the remaining substance, called an absolute, is a very concentrated aromatic compound. Absolutes are widely used within the botanical perfume and cosmetic fields.
Types of Solvent Extraction:
 Again, some plant materials, like vanilla, which is difficult to extract, or jasmine flowers or rose petals, which are too delicate to survive the process of distillation, use another method to extract their aromatic essence, a process called solvent extraction.
Again, some plant materials, like vanilla, which is difficult to extract, or jasmine flowers or rose petals, which are too delicate to survive the process of distillation, use another method to extract their aromatic essence, a process called solvent extraction.
This process utilizes petroleum solvents such as petroleum ether, hexane or toluene; alcohol solvents such as methanol or ethanol; or carbon dioxide.
As the solvent is added to the botanical it is absorbed and allows the release of the aromatic compounds.
These solvent-extracted compounds are called "absolutes" and are very concentrated. Technically they can not be called essential oils.
They are either extracted using:
- petroleum solvents (like hexane)
- ethanol solvents
- supercritical CO2 extraction process. This method helps to maintain the integrity of the base constituents of the oil and produces a highly concentrated finished product without using petroleum solvents.
Solvent extraction involves soaking plant material in a solvent to draw the fragrance out. It works for any type of plant matter but is typically used for more fragile material, like flowers. The resulting mixture is then processed several times to separate the absolute from the solvent and any plant residue.
Petroleum Solvent: A few years ago almost all absolutes, essential oils, and even carrier oils were extracted in this manner. Since this process allows companies to capture more of the essential oil components, it allows companies to get more bang for their buck.
However, since residues of the petroleum solvent, like hexane or benzene, may remain in the aromatic oil, solvent extraction was not recommended for essential oils used for aromatherapy. These residues may also cause allergic reactions and skin irritation.
Ethanol Solvent: While petroleum solvents can be very dangerous, ethanol (food-grade drinking alcohol) is a rather benign solvent. The plant material is steeped in ethanol. The alcohol is evaporated out and a highly concentrated oil known as an absolute is left behind. Sometimes people will list an absolute, like vanilla, as an essential oil, but absolutes are not technically essential oils.
Supercritical Carbon Dioxide Extraction: Carbon dioxide extraction is steadily gaining popularity as a more natural method to extract the essential oil from botanical material. CO2 extraction uses carbon dioxide as the solvent to produce something called an extract.
Unlike traditional solvent extraction, however, no residue is left behind which generally makes the resulting oils purer and safer for use.

This method produces nice aromatic oils but is also more expensive.
Supercritical CO2 extraction have several advantages over other extraction methods. The processes occurs at lower temperatures than steam distillation, thereby preserving delicate volatile constituents.
Carbon Dioxide is colorless, odorless, flavorless, and non-toxic. It is considered an excellent solvent since carbon dioxide does not chemically interact with the essential oil extracted. The oils produced have a longer shelf life, and the extraction process is environmental friendly than using petroleum solvents.
Under pressure, the temperature of the carbon dioxide is increased to about 92 degrees Fahrenheit (33 degrees Celsius). At this temperature carbon dioxide enters a “supercritical” phase, meaning that is part liquid and part gas.
To remove the carbon dioxide solvent, simply decrease the pressure and the carbon dioxide returns to a gaseous state leaving behind a pure essential oil-like compound with no residues. While close to an essential oil, these extracts are still not considered to be 100% pure essential oils.
Since CO2 extraction takes place in a completely sealed chamber, it recovers the whole oil from the plant material, including any pesticide residue. Thus, the resulting oil may have a greater amount of pesticides in it than other conventional extraction methods. For this reason we recommend organic CO2 extracts only.
The Organic Rosemary Oil Extract we use as an antioxidant in some of our products is extracted using this Carbon Dioxide method. It is an extraction method that can be used to obtain a USDA Certified Organic Certification.
Conclusion
While essential oils, CO2 Extracts and absolutes are all considered aromatic extracts, what makes them unique is their method of extraction and the resulting chemical composition of each type of extraction.
In order to benefit from the therapeutic properties it is important to choose purely pure oil, distilled with no additives and dangerous solvents. Choosing certified organic oils is a great place to begin.
The most important consideration when purchasing essential oils is to buy from a reputable supplier that lists the method of extraction and tests their oils for the presence of adulterated additives.
When purchasing an essential oil or a product containing essential oils, be sure to ask about the extraction method!
The essential oils used in Chagrin Valley products are steam distilled except for the citrus oils which are cold-pressed.

Why We Use Only Real Plant Essential Oils?
How to Diffuse Essential Oils for Personal Use
Why Are Synthetic Fragrance Oils So Popular?
Natural Fragrance Oil? . . . Really?


